
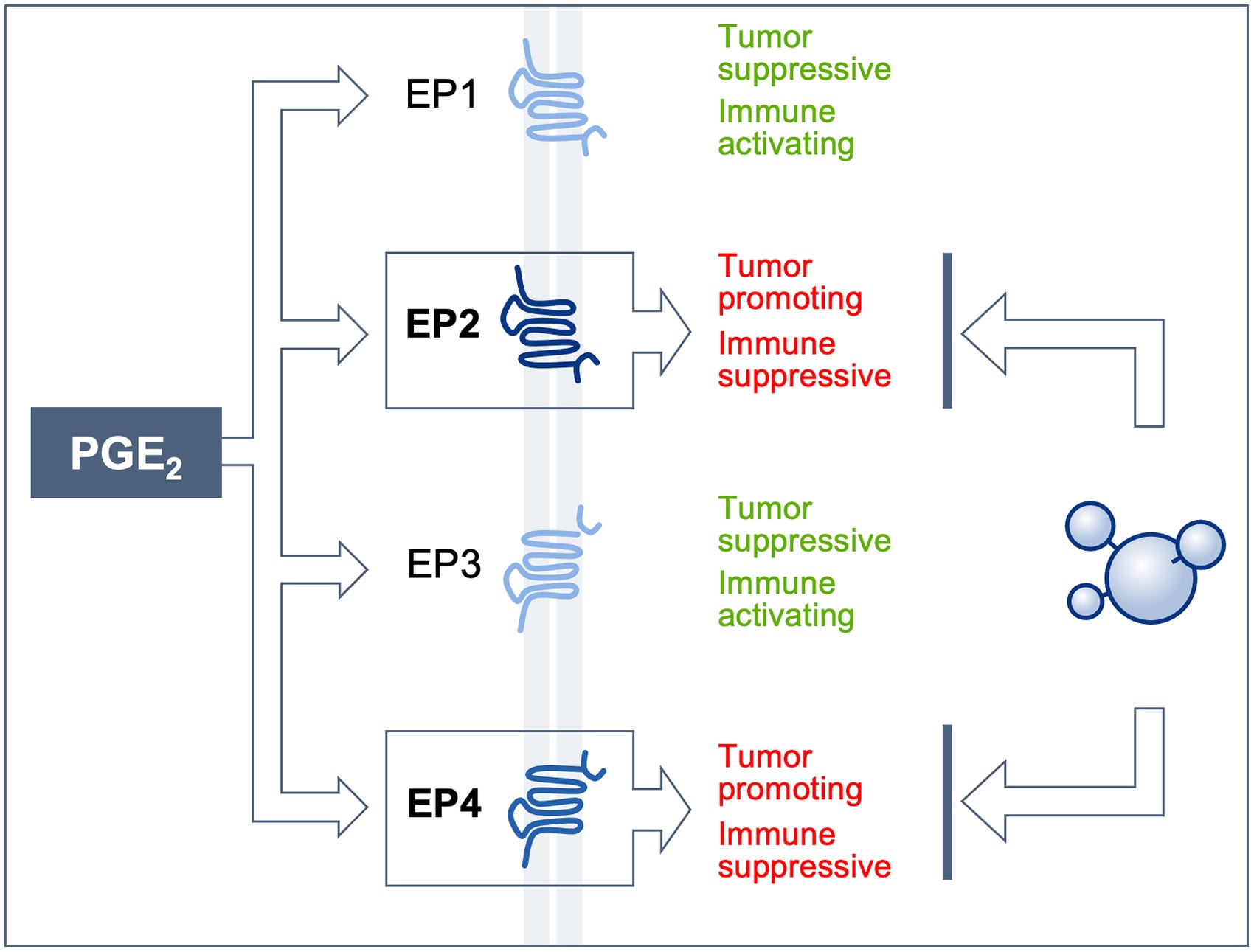
Tempest is headquartered in South San Francisco and supported by notable healthcare investors. Tempest is also developing an inhibitor of TREX-1, an exonuclease highly expressed in tumors that suppresses both STING and tumor immunity, to activate STING selectively in tumors via a systemically-delivered small molecule. Both TPST-1120 and TPST-1495 are progressing through Phase 1 studies designed to study both agents as monotherapies and in combination with other approved agents. The company’s two novel clinical programs are TPST-1120 and TPST-1495, antagonists of PPARα and EP2/4, respectively. Tempest Therapeutics is a clinical-stage oncology company advancing small molecules that combine both precision and immune-mediated mechanisms with the potential to target a wide range of tumors. To access the archived recording of the company presentation, please visit the Tempest website at About Tempest Therapeutics The presentation will be available on Monday, Januat 6:00 am Eastern Time. Wainwright Virtual BioConnect Conference. 08, 2021 (GLOBE NEWSWIRE) - Tempest Therapeutics, Inc., a clinical-stage oncology company developing first-in-class therapeutics that combine both precision and immune-mediated mechanisms, today announced that Tom Dubensky, Ph.D., chief executive officer, and Steve Brady, president and chief operating officer, will present at the H.C. Michelle Dipps Tempest Therapeutics shares are currently valued. Michelle Dipp is the largest individual Tempest Therapeutics shareholder, owning 7.96M shares representing 77.10 of the company. was founded in 2003 and is headquartered in Montreal, Canada.SOUTH SAN FRANCISCO, Calif., Jan. Tempest Therapeutics (NASDAQ: TPST) is owned by 20.35 institutional shareholders, 495.77 Tempest Therapeutics insiders, and 0.00 retail investors. (TPST)s stock analysis, price valuation, corporate actions, and financials from Indias independent mutual fund research. and development agreement with Cynapsus Therapeutics Inc. The company has licensing, development, and supply agreement with Tilray, Inc. It offers INT0008/2008, a Rizatriptan oral film product for the treatment of migraine INT0046/2018 for adult use INT0007/2006, an oral film product for treatment of erectile dysfunction INT0043/2015, an oral film containing montelukast for treatment of Alzheimer INT0027/2011 to treat opioid addition INT0010/2006 for treatment of neuropathic pain and nausea in cancer patients undergoing chemotherapy INT0036/2013, an oral film product for schizophrenia or bipolar 1 disorder and INT0048/2020 for animal health. 
IntelGenx Technologies Corp., a drug delivery company, focuses on the development and manufacturing of novel oral thin film products for the pharmaceutical market.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
